


personalization of care



personalization of care
Team

CEO
Chairwoman
MORE INFO...
Véronique Foutel, Pharmaceutical Doctor, former Paris Hospital Resident (CEO) has 30+ years of experience dedicated to Life Science innovation, encompassing 16 years within the Pharma Division of F. Hoffmann La Roche AG.for which she co-led and led Strategic Pricing function for new products from 2004 to 2009. Veronique used to a strategic advisor to Pharma / biotech management Teams since 2012 before taking the helm of InBrain Pharma in 2024. She holds a pharmaceutical sciences doctorate from Paris Cité University, and is a former Paris Hospital Resident.

Dr. Matthieu Fisichella
CSO
Co-founder
MORE INFO...
Matthieu Fisichella, PhD (CSO and Co-founder) is a Doctor-Engineer in Biology and Challenge+ at HEC. Former CEO of InBrain Pharma from 2018 to 2023, he has previously been project manager in life sciences at SATT Nord and scientific and partnership manager at Primadiag. Matthieu has evaluated and structured 10 projects, detected 55 technologies, obtained and managed +€2m of budget, and transferred 2 projects.

Pr. David Devos
Scientific and Clinical Advisor
Co-founder
MORE INFO...
David Devos, MD, PhD (Preclinical and clinical scientific advisor, Co-founder) is the Director of Neuroscience research team DVCD INSERM UMRS-U1172 as well as a Neurologist, Professor of Medical Pharmacology, Faculty of Medicine & Neurologist University Hospital. He has been an executive board member of the Parkinson network Ns-Park and is author/coauthor of 366+ publications including 250+ articles, H-Index > 60. He has been involved in 31 clinical studies including 14 multicenter studies and 14 as coordinator (10 in progress), in 5 academic patent filing.

Pr. Caroline Moreau-Devos
Clinical Scientific Advisor
Co-founder
MORE INFO...
Caroline Moreau-Devos, MD, PhD (clinical scientific advisor, Co-founder) is Professor of Neurology at Lille University hospital, assistant secretary of the French Society of Neurology, French representative at the European Academy of Neurology, author / co-author of 130+ articles, awarded with the Fondation Recherche Médicale Prize (2022) has contributed to 34 clinical studies including 14 multicenter studies and 5 as coordinator, has filed 5 patents and has been key into 2 industrial transfers.
Scientific Advisory Board

Pr. Alberto Espay
MORE INFO...
He is a neurologist specializing in movement disorders, with more than 400 scientific articles, 40 book chapters, and 10 books to his credit—including Common Movement Disorders Pitfalls and Brain Fables.
Trained at Indiana University in neurology and at the University of Toronto in the electrophysiology of movement disorders, holding an MSc in clinical epidemiology, he brings dual clinical and methodological expertise.
His research focuses on the development of biomarkers that are “agnostic” to traditional clinical diagnoses to better target therapies for neurodegenerative diseases.

Pr. Alfonso Fasano
MORE INFO...
He is co-director of the Movement Disorders Surgery Program at Toronto Western Hospital and a neurologist at The Hospital for Sick Children in Toronto.
As a clinician-scientist at the Krembil Research Institute and affiliated with the KITE Center at the Toronto Rehabilitation Institute, he leads the closed-loop component of the CRANIA (Center for Advancing Neurotechnological Innovation to Application) program.
His scientific interests include:
• the use of advanced technologies (infusion pumps, neuromodulation) in movement disorders,
• the pathophysiology of tremors and gait disorders,
• the study of neural circuits and their translation into innovative clinical applications.

Pr. Per Odin
MORE INFO...
A renowned specialist in movement disorders—particularly Parkinson’s disease—he is the author of more than 170 scientific publications and leads numerous research projects on advanced therapies and non-motor symptoms.

Pr. Olivier Rascol
MORE INFO...
A specialist in the neuropharmacology of movement disorders, his work focuses on the development of dopaminergic and non-dopaminergic treatments, neuroprotection, and functional imaging.

Dominic Benning
MORE INFO...
He leads the dopaminergic therapy franchise of EverPharma at the Headquarters in Austria. He has a thorough understanding of Parkinosn’s Disease Market and its key players.
Board

Dr. Véronique Foutel
CEO
Chairwoman
MORE INFO...
Véronique Foutel, Pharmaceutical Doctor, former Paris Hospital Resident (CEO) has 30+ years of experience dedicated to Life Science innovation, encompassing 16 years within the Pharma Division of F. Hoffmann La Roche AG.for which she co-led and led Strategic Pricing function for new products from 2004 to 2009. Veronique used to a strategic advisor to Pharma / biotech management Teams since 2012 before taking the helm of InBrain Pharma in 2024. She holds a pharmaceutical sciences doctorate from Paris Cité University, and is a former Paris Hospital Resident.

Dr. Matthieu Fisichella
CSO
Co-founder
MORE INFO...
Matthieu Fisichella, PhD (CSO and Co-founder) is a Doctor-Engineer in Biology and Challenge+ at HEC. Former CEO of InBrain Pharma from 2018 to 2023, he has previously been project manager in life sciences at SATT Nord and scientific and partnership manager at Primadiag. Matthieu has evaluated and structured 10 projects, detected 55 technologies, obtained and managed +€2m of budget, and transferred 2 projects.

Pr. David Devos
Scientific and Clinical Advisor
Co-founder
MORE INFO...
David Devos, MD, PhD (Preclinical and clinical scientific advisor, Co-founder) is the Director of Neuroscience research team DVCD INSERM UMRS-U1172 as well as a Neurologist, Professor of Medical Pharmacology, Faculty of Medicine & Neurologist University Hospital. He has been an executive board member of the Parkinson network Ns-Park and is author/coauthor of 366+ publications including 250+ articles, H-Index > 60. He has been involved in 31 clinical studies including 14 multicenter studies and 14 as coordinator (10 in progress), in 5 academic patent filing.

Pr. Caroline Moreau-Devos
Clinical Scientific Advisor
Co-founder
MORE INFO...
Caroline Moreau-Devos, MD, PhD (clinical scientific advisor, Co-founder) is Professor of Neurology at Lille University hospital, assistant secretary of the French Society of Neurology, French representative at the European Academy of Neurology, author / co-author of 130+ articles, awarded with the Fondation Recherche Médicale Prize (2022) has contributed to 34 clinical studies including 14 multicenter studies and 5 as coordinator, has filed 5 patents and has been key into 2 industrial transfers.

Thomas Bexant
Board Member
Finovam Gestion
MORE INFO...
Thomas Bexant is a member of the executive board of Finovam Gestion, a venture capital firm supporting technology start-ups in the Hauts-de-France, Grand Est and Bourgogne-Franche-Comté regions.
With specialized training in biotechnology and agri-food, he has over 20 years of experience in innovation financing, private equity, and entrepreneurship. He has managed several companies and supported over 50 growth projects.
At Finovam, he brings strategic and operational vision to the innovative companies he supports, particularly in the life sciences, greentech, and deeptech sectors.

Guillaume Bruniaux
Board Member
Nord France Amorçage
MORE INFO...
Guillaume Bruniaux is a director at Siparex and oversees the management of the Nord France Amorçage fund, which is dedicated to financing innovative companies in the Hauts-de-France region.
With over 15 years of experience supporting regional leaders and projects, he puts his expertise to work for startups in the launch or seed phase.
Collaborations
MORE INFO...
Infusyn Therapeutics, LLC, based in Hackettstown, NJ (USA), manufactures and markets the Prometra II® pump, the most sophisticated and precise implantable, fully-programmable drug delivery pump on the market. Prometra is approved by the US FDA for intrathecal delivery of opioids for the treatment of pain and baclofen for the treatment of spasticity. InBrain utilized the Prometra II® pump for intracerebroventricular infusion of A-dopamine in the DIVE I clinical trial conducted between 2020 and 2025. Infusyn is also developing Prometra with its partners in oncology, CNS and cardio-pulmonary indications where precision dosing is crucial for improved efficacy and better patient outcomes.
MORE INFO...
IDD-Xpert will support InBrain Pharma’s efforts in setting and scaling up a GMP compliant production line for A-dopamine under sterile and very strict anaerobic conditions.
MORE INFO...
InBrain Pharma is a spin-off of the University of Lille, born out of academic and scientific excellence. InBrain Pharma works closely with the Lille Neuroscience & Cognition Research Center (LilNCog – INSERM CHU Lille- ULille), renowned for its multidisciplinary expertise in fundamental, clinical, and translational neuroscience. High-level scientific expertise, cutting-edge research infrastructure and an academic ecosystem that stimulates innovation in the field of brain health are made accessible through this proximity.
MORE INFO...
A-dopamine for advanced Parkinson’s disease management : a medical innovation born at Lille University Hospital (CHU de Lille)
The A-dopamine project was initiated at CHU de Lille thanks to the work of two renowned neurology specialists, Prof. David Devos and Prof. Caroline Moreau. Together, they developed a new therapeutic approach protected by two patents, for which CHU de Lille is co‑owner.
To accelerate its development, CHU de Lille authorized the two researchers to co‑found the start‑up IBP, where they contribute their scientific expertise.
A public–private partnership to advance research for the benefit of patients
InBrain Pharma and CHU de Lille worked hand-in-hand to carry out the DIVE‑I monocentric clinical trial, conducted within the neurology department of the hospital.
In this context:
- CHU de Lille handled all regulatory procedures required to launch the study.
- It ensured daily operational oversight, including monitoring, data management, and budget follow‑up.
- The investigational drug, A‑dopamine, was developed and manufactured at CHU de Lille, in connection with the two patents underlying the DIVE technology.
- CHU teams also performed the statistical analyses necessary to interpret the results.
A shared ambition
This partnership between a major healthcare institution and an innovative young company demonstrates how medical expertise and entrepreneurial agility can complement each other to accelerate the availability of new solutions for patients.
Technology
Parkinson’s disease
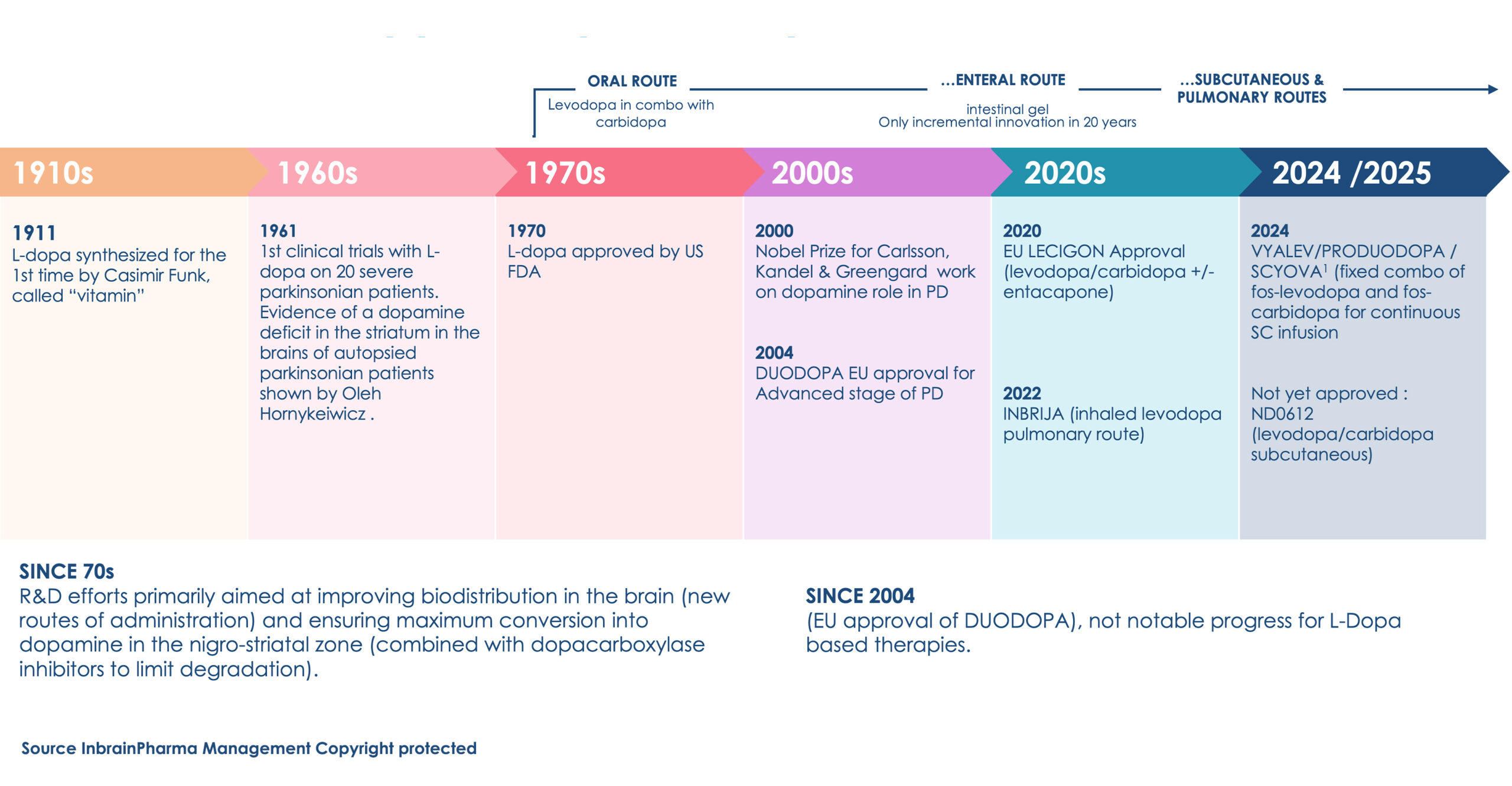
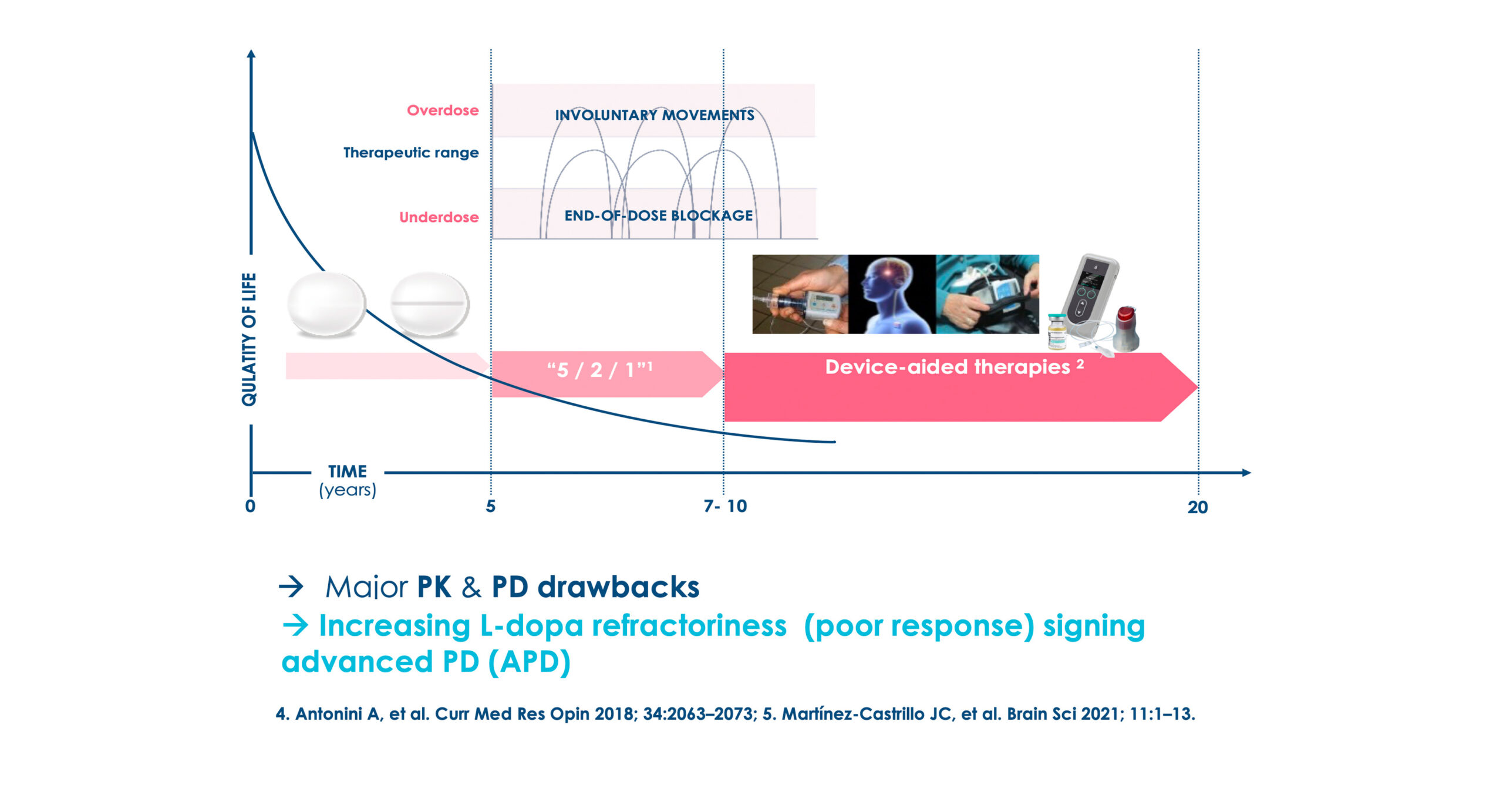
Backbone Therapy: L-dopa for 55 years
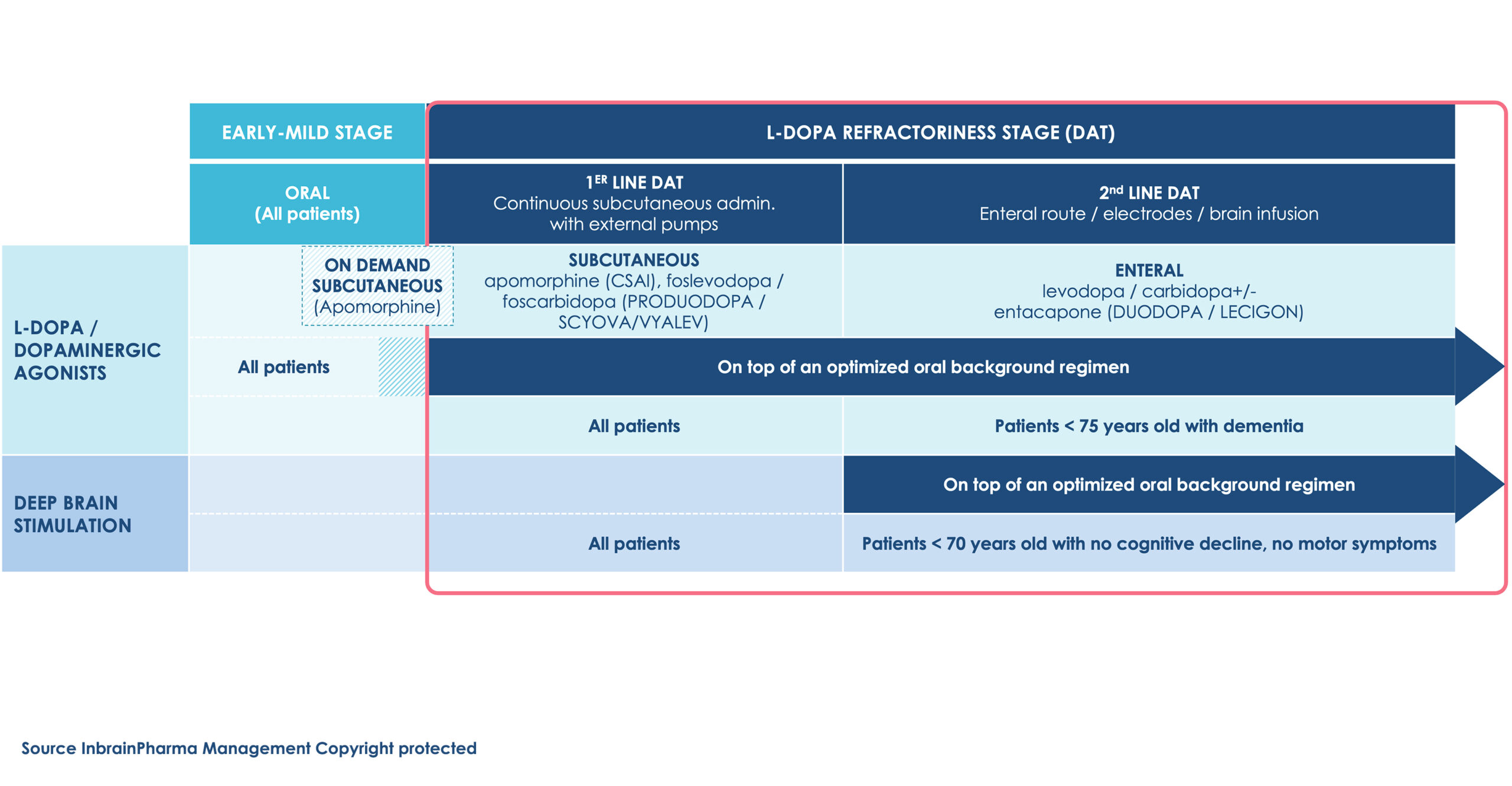
Current Device Aided Therapies (DAT) in advanced stage
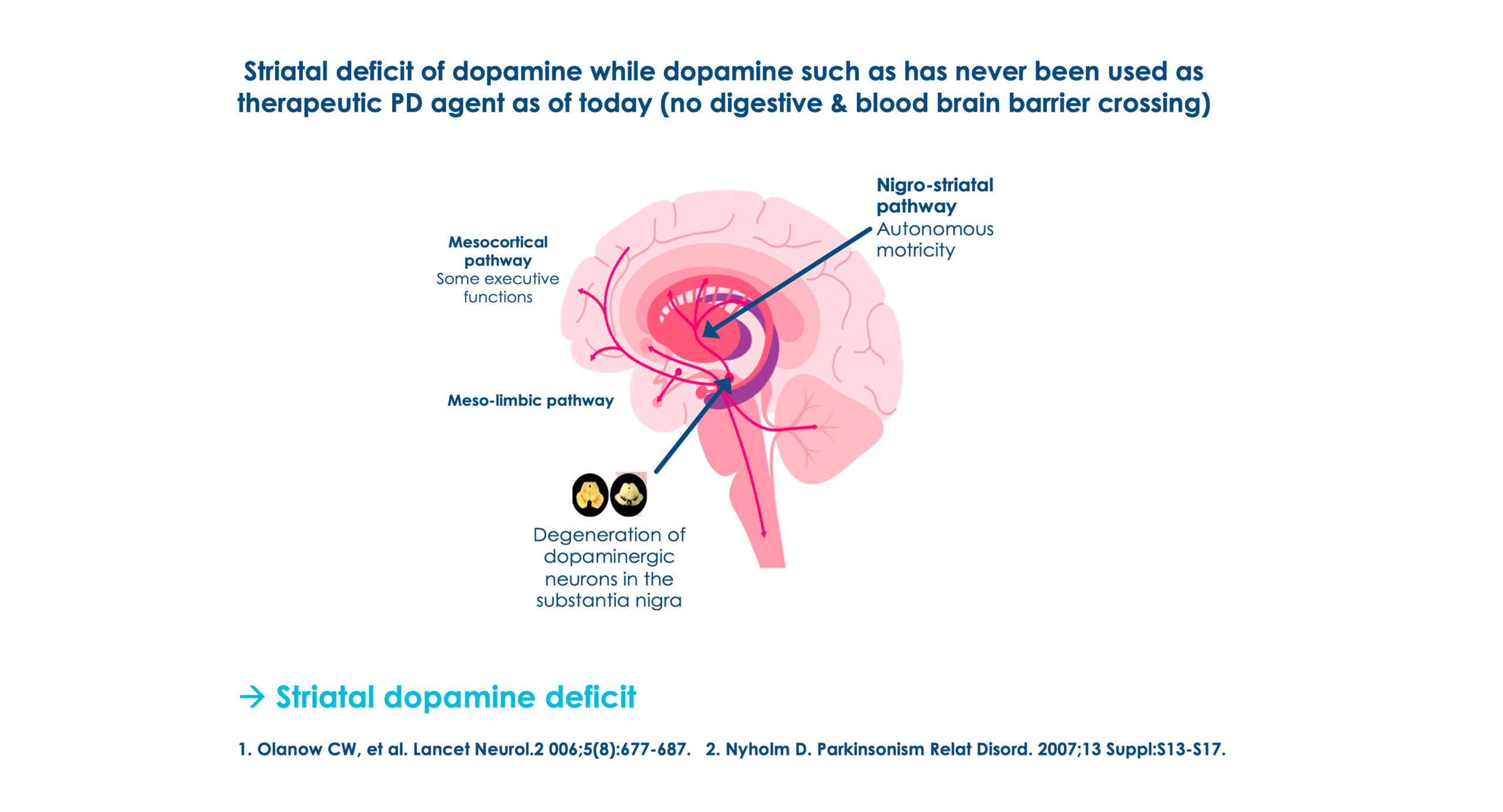
Parkinson’s Disease (PD) Etiology
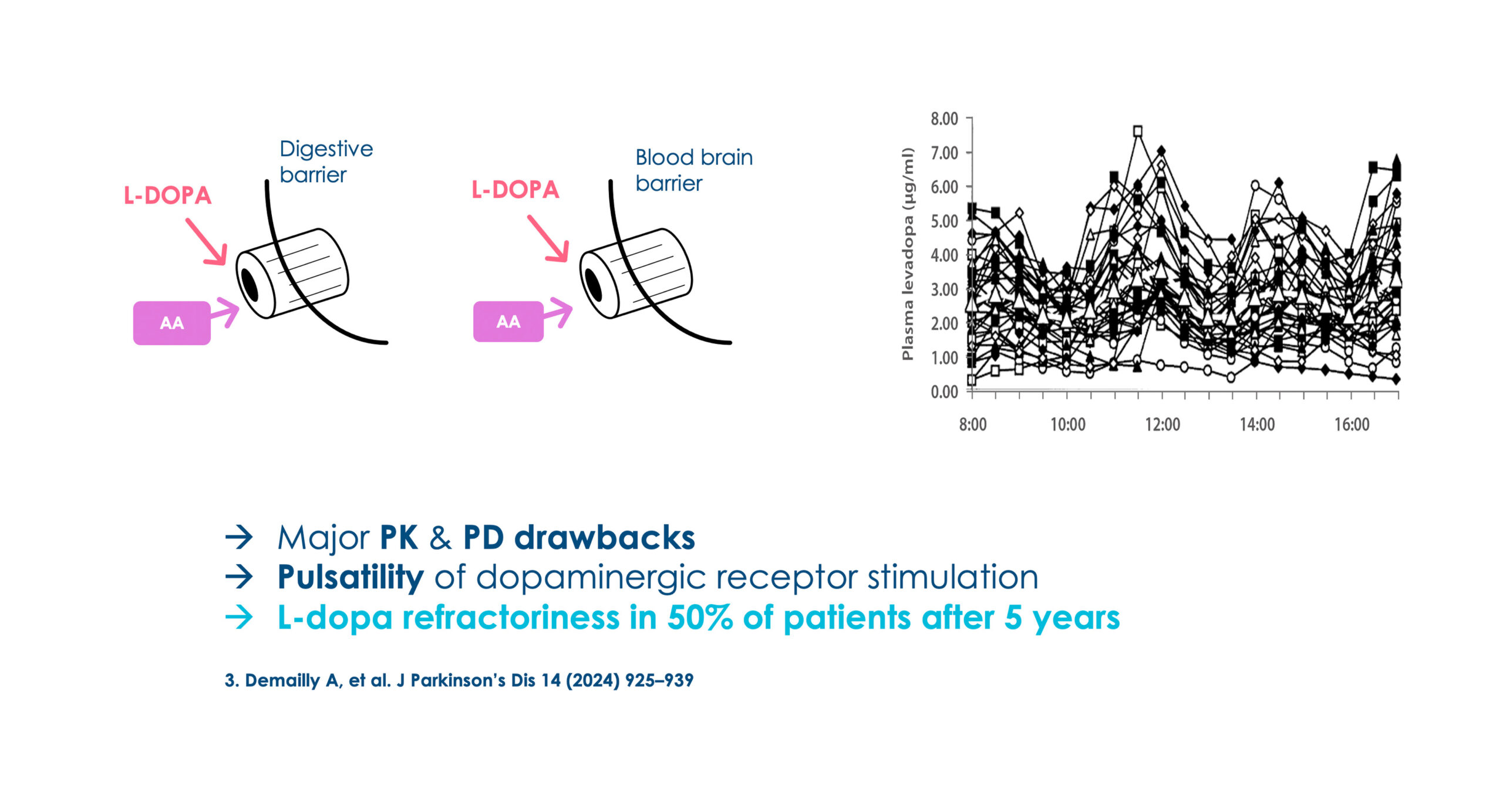
L-dopa: Backbone PD therapy
L-dopa based Device-Aided Therapies (DAT)
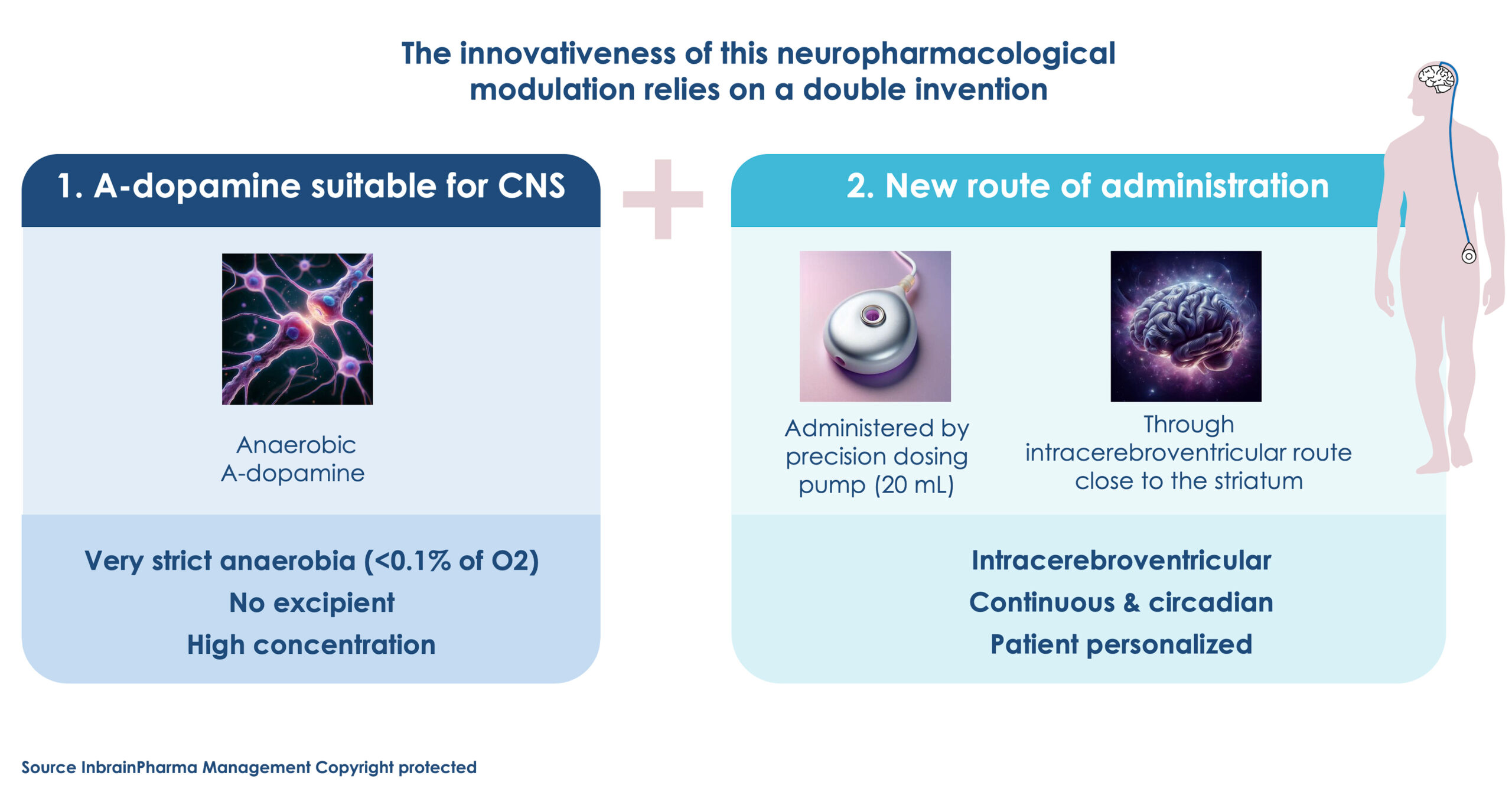
A-Dopamine
An innovative therapy for pd patients refractory to L-dopa treatments
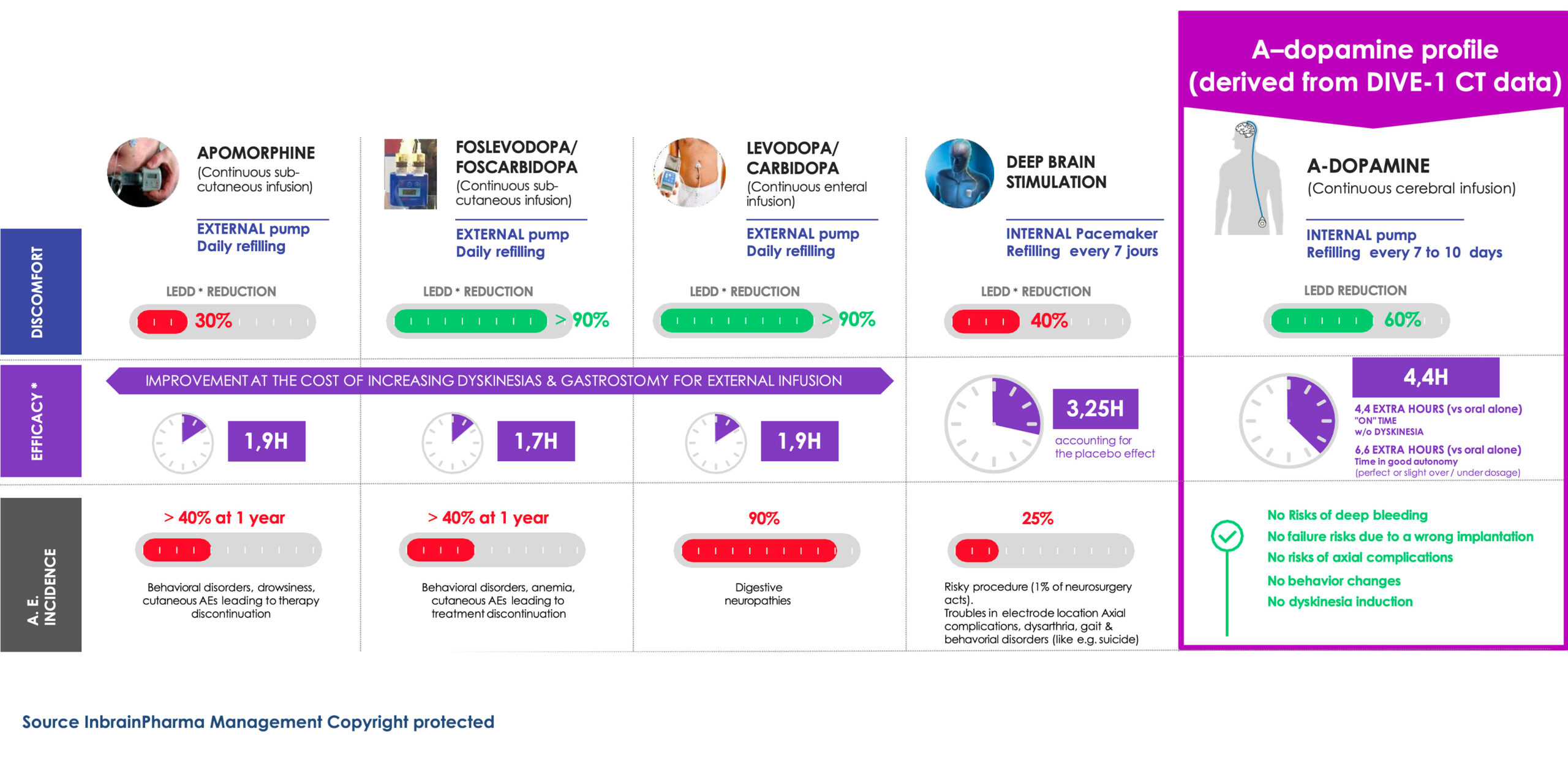
Overview of the current DAT market
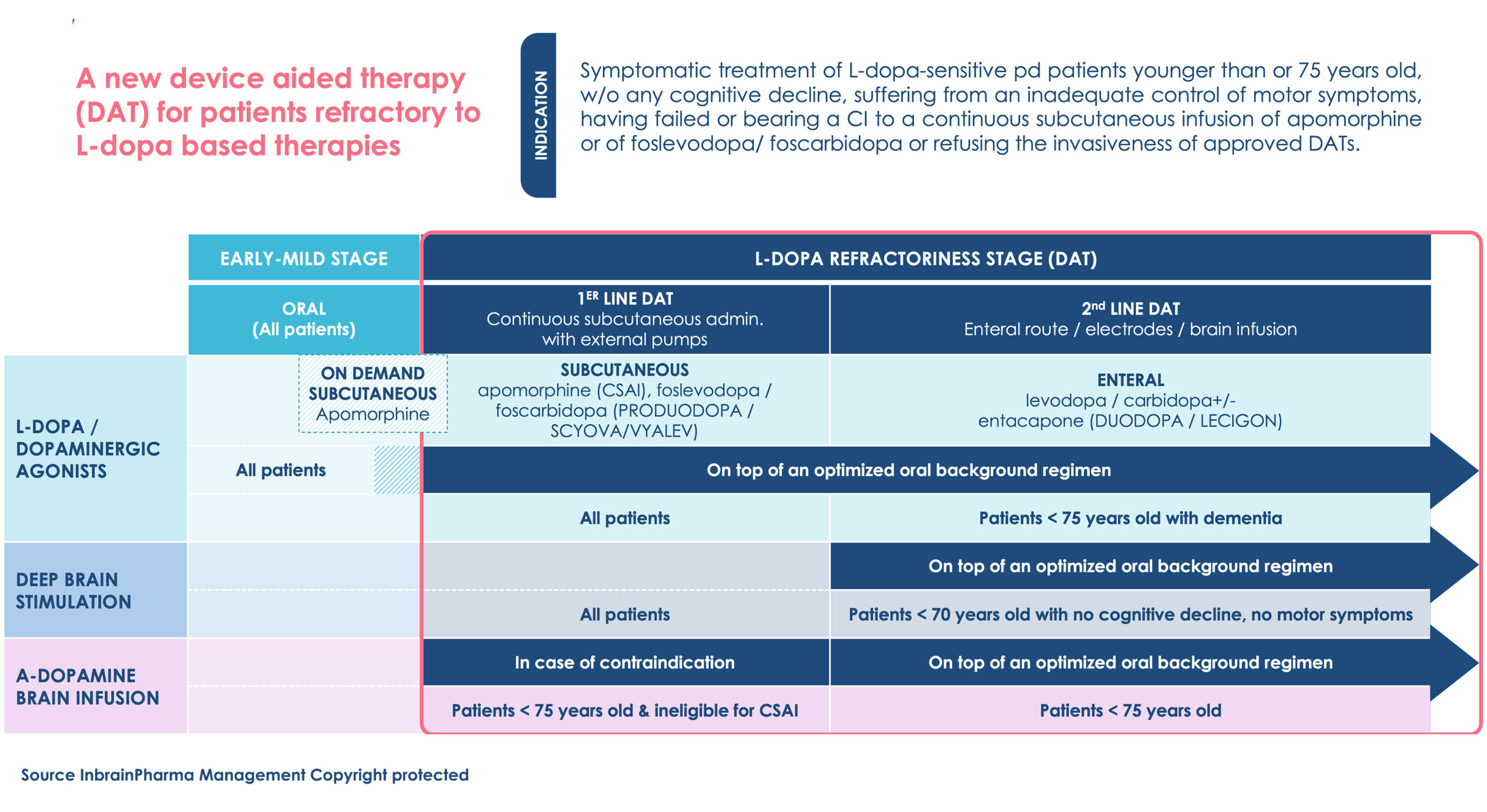
Indication & positioning (EMA endorsed)
A solution addressing a high unmet need

Pipeline




Investors
MORE INFO...
Finovam Gestion is an approved management company based in the Hauts-de-France, Grand Est, and Bourgogne-Franche-Comté regions. It supports innovative companies with high potential from the earliest stages (seed funding). Its preferred sectors are information and communication technologies (ICT/IT), health and biotechnology, bioeconomy/agro-resources, industry of the future, and energy transition. With strong regional roots, Finovam Gestion leverages its proximity to research centers, incubators, and manufacturers in northeastern France to identify and support future technology leaders.
MORE INFO...
Since 2013, Nord France Amorçage (NFA) has been supporting innovative start-ups in the Hauts-de-France region during their seed phase. The fund, which is wholly owned by the Hauts-de-France Region with the support of European funds, co-invests alongside players such as Finovam Gestion.
MORE INFO...
The University of Lille Foundation, a local investor committed to fostering innovation, supports our development as part of its mission to promote research and scientific excellence.
As a partner foundation of the University of Lille, it notably supports entrepreneurial projects with a strong technological or scientific component, originating from Lille’s academic research, thereby contributing to value creation in the region. By investing in InBrain Pharma, the University of Lille Foundation reaffirms its role as a key player in technology transfer.
MORE INFO...
France Parkinson, a non-profit organization since 1988, has been working since 1984 to support people with Parkinson’s disease, assist their loved ones, train professionals, and fund research. By taking an equity stake in InBrain Pharma company, France Parkinson has showed a strong commitment to innovation and sourcing therapeutic breakthroughs for the disease management. This demonstrates its confidence in InBrain Pharma vision, expertise, and ability to make a real impact on patient’s life.
MORE INFO...
Bpifrance, the French public investment bank, supports our development through several strategic initiatives dedicated to innovation and industrialization. As winners of the i-Lab innovation competition and Deeptech-certified, we benefit from Bpifrance’s support in transforming a disruptive technology from research into a high-impact industrial project.
This partnership is also part of the France 2030 – “Première Usine” national program, which supports reindustrialization and production in France. Thanks to this support, InBrain Pharma can accelerate its industrial scale up, while contributing to France’s technological sovereignty.
MORE INFO...
The Region Hauts-de-France supports our company through its programs dedicated to research, development, and innovation (RDI). This regional commitment supports the day-to-day R&D activities of InBrain Pharma.

InBrainPharma is currently open to collaboration with industrial and/or financial partners on the next phases of its development, please contact us.
News & events
Press Releases
Careers
There are currently no open positions in the InBrainPharma team.











